As the holiday surge of COVID-19 infections declines in Chicago’s Black neighborhoods, the Food and Drug Administration on Monday, January 23, proposed once-a-year COVID-19 shot for most adults and children.
Health officials aim to make COVID-19 vaccinations more like the annual flu shot.
This week, the FDA will ask its panel of outside vaccine experts to weigh in on the proposal. The agency is expected to take their advice into consideration while deciding future vaccine requirements for manufacturers.
According to posted documents online, FDA scientists say many Americans now have “sufficient pre-existing immunity” against the coronavirus because of vaccination, infection or a combination of the two.
Experts say that layer of protection should be enough to move to an annual booster against the latest Omicron strains and make COVID-19 vaccinations more like the yearly flu shot, according to the agency.
However, experts say a two-dose combination may be needed to protect adults with weakened immune systems and very small children. FDA scientists and vaccine companies would study vaccination, infection rates and other data to decide who should receive a single shot versus a two-dose series.
The FDA also plans to ask the panel to vote on whether all vaccines should target the same strains. That step would be needed to make the shots interchangeable, doing away with the current complicated system of primary vaccinations and boosters.
More than 80 percent of the U.S. population has had at least one vaccine dose, only 16 percent of those eligible have received the latest boosters authorized in August.
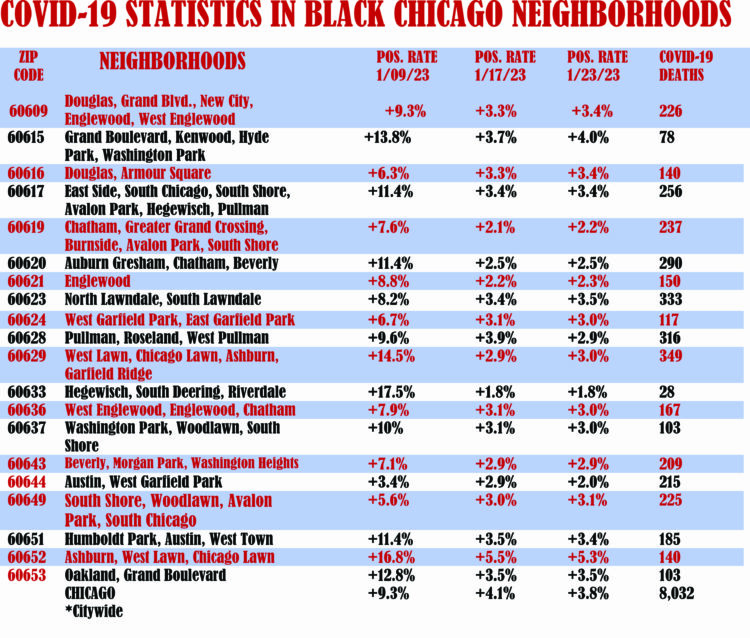
For the second straight week, the positivity rates in Chicago have dropped as the city’s average fell to 3.8 percent as of January 23. Last week, Chicago’s positivity rate was 4.1 percent.
Of 20 Black zip codes, only two (60615 and 60653) were higher than the city’s average of 3.8 percent. Zip code 60615, which includes Grand Boulevard, Washington Park, Hyde Park and Kenwood, was 4.0 percent. Zip code 60653, which includes Ashburn, West Lawn and Chicago Lawn, was 5.3 percent.
According to the latest Community Level metrics by the Centers for Disease Control and Prevention (CDC), Chicago and Cook County remain at the Medium COVID-19 Community Level. In all, 74 of Illinois’ 102 counties were at the low level this week (43 last week), while 25 were medium (56 last week), and three were high (three last week).
“No one is more pleased than I am that Chicago remains in the Medium Level,” said CDPH Commissioner Allison Arwady in a statement.
“I think this is evidence that Chicagoans are doing the right thing—wearing masks in public settings, staying home when they are sick, and even cracking windows to improve ventilation when gathering.”
“But we remain prepared in case we do move to the High Level in coming weeks. While the new Omicron subvariant XBB 1.5 remains less than 25 percent of cases in the Midwest, it is nearly 50 percent of cases across the U.S.
“This variant appears to be the most transmissible yet, but the updated COVID-19 booster vaccine works well against it. One more reason for everyone to stay up to date on vaccines and get their new booster.”
The initial shots from Pfizer and Moderna, which are called the primary series, target the strain of the virus that first emerged in 2020 and quickly swept across the world. The updated boosters launched last fall were also tweaked to target Omicron relatives that had been dominant.
Under the FDA’s proposal, the agency, independent experts and manufacturers, would decide annually on which strains to target by the early summer, allowing several months to produce and launch updated shots before the fall.
Ultimately, FDA officials say moving to an annual schedule would make it easier to promote future vaccination campaigns, which they hope would boost vaccination rates nationwide in the future.
The original two-dose COVID shots have offered strong protection against severe disease and death no matter the variant, but protection against mild infection wanes. Experts continue to debate whether the latest round of boosters significantly enhanced protection, particularly for younger, healthier Americans.